Document Lifecycle Management
Manage creation, approval, and controlled distribution of SOPs, batch records, policies, and other critical documents.

Helyx Controlled Documents
Streamlined document management while ensuring full traceability and compliance, helping life sciences companies improve efficiency and regulatory readiness.

Manage creation, approval, and controlled distribution of SOPs, batch records, policies, and other critical documents.
Streamline approvals, reviews, and notifications with automated routing to the right stakeholders.
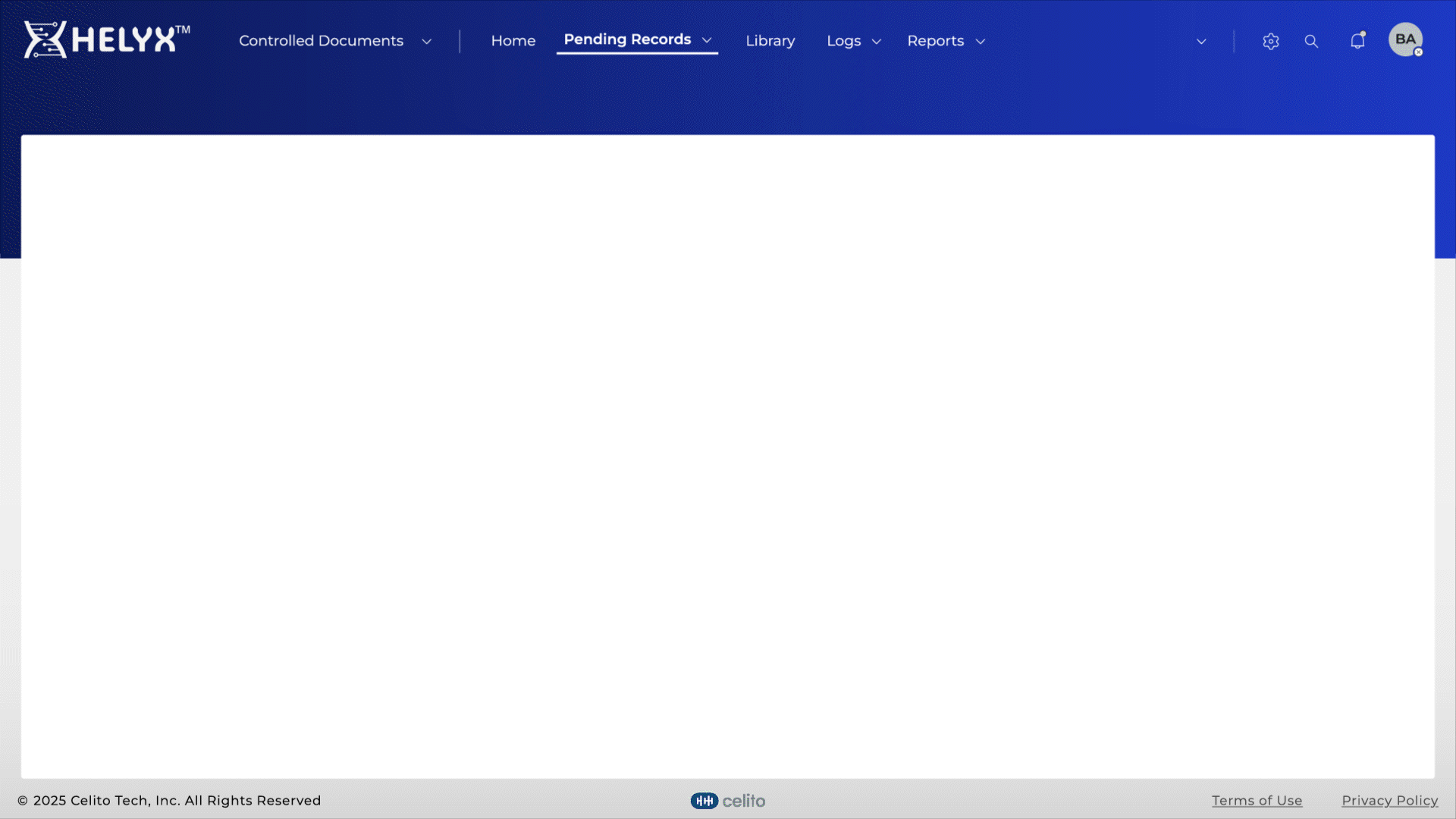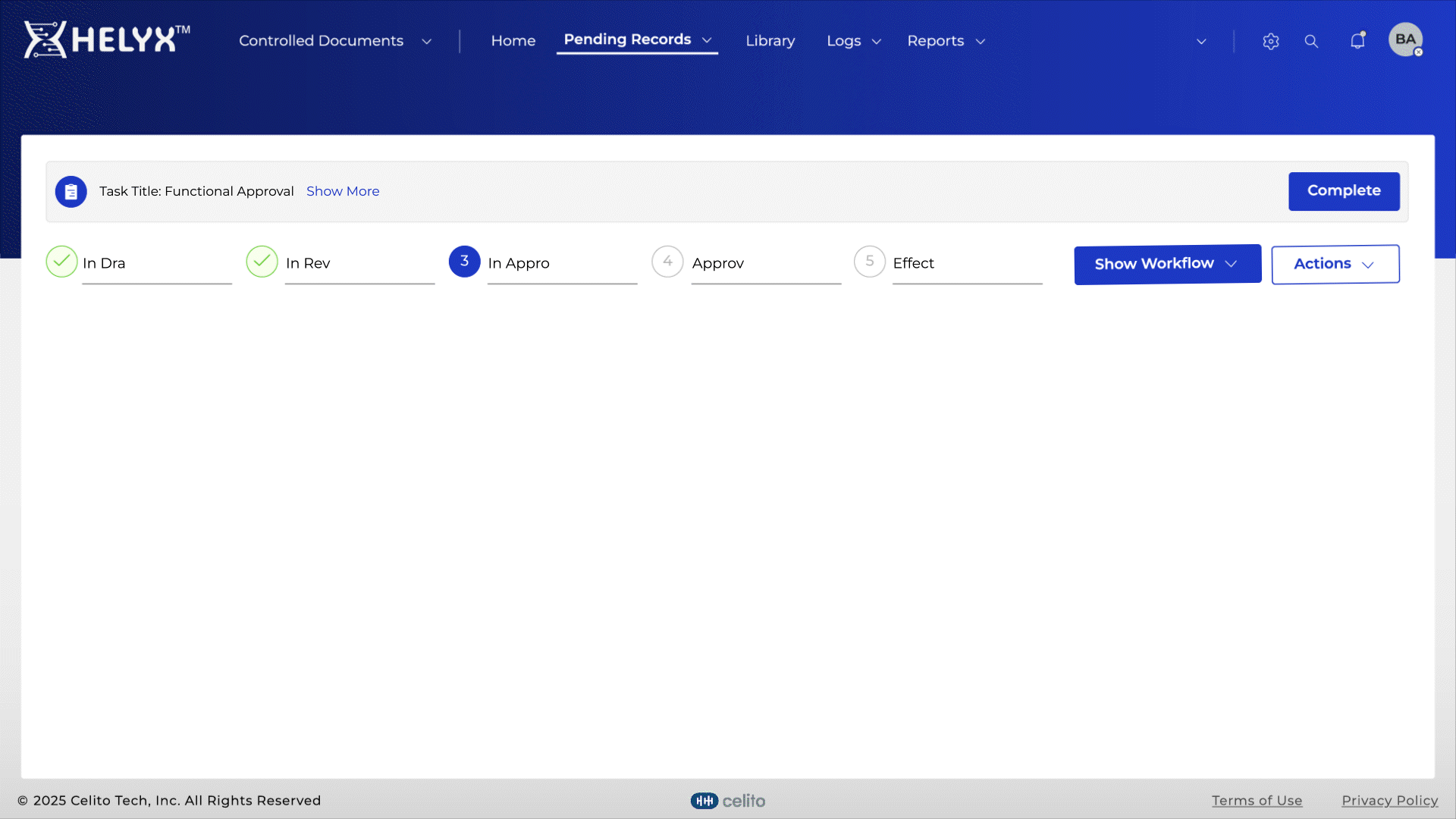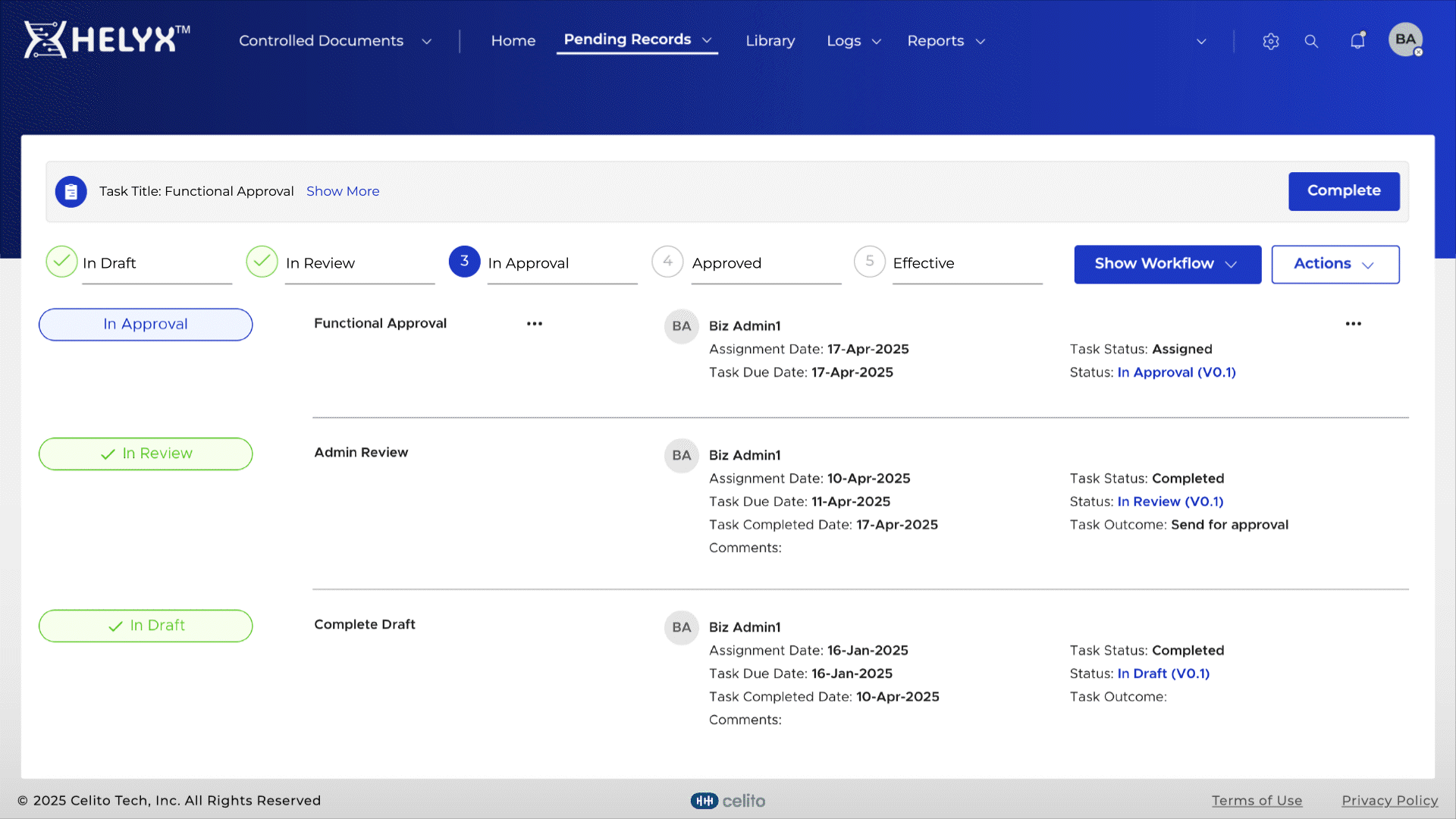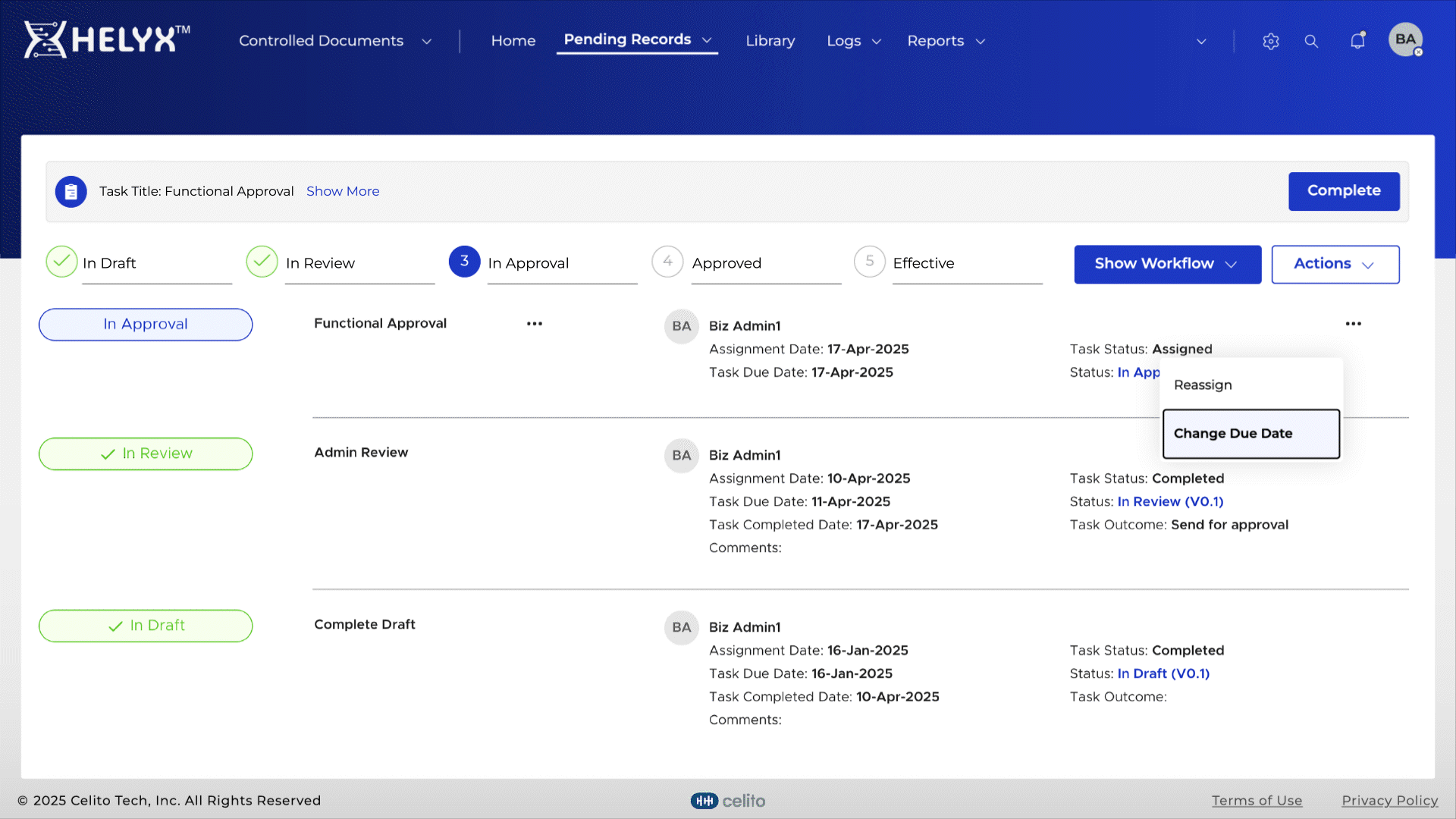
Automatically maintain previous document versions for complete traceability.
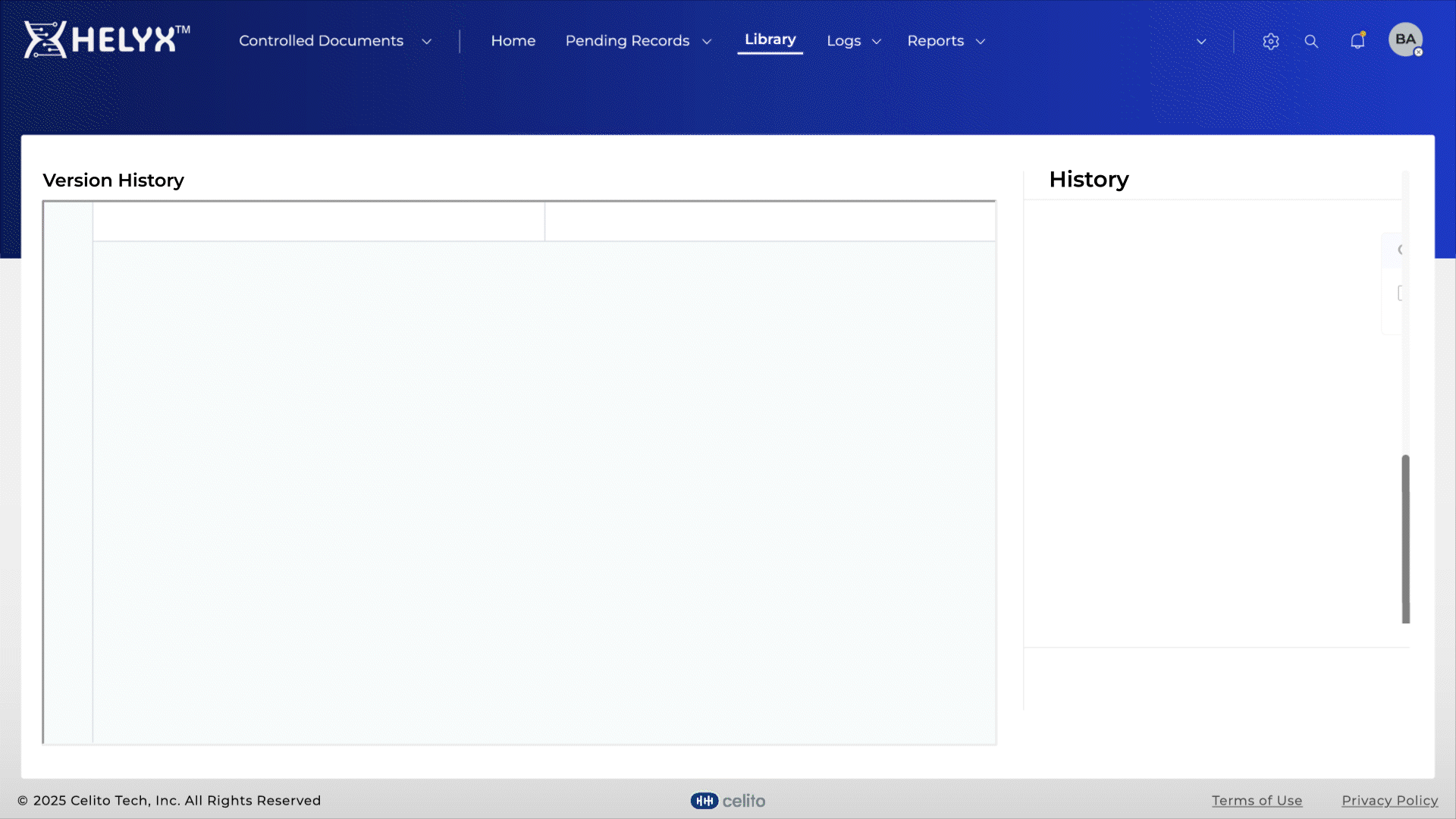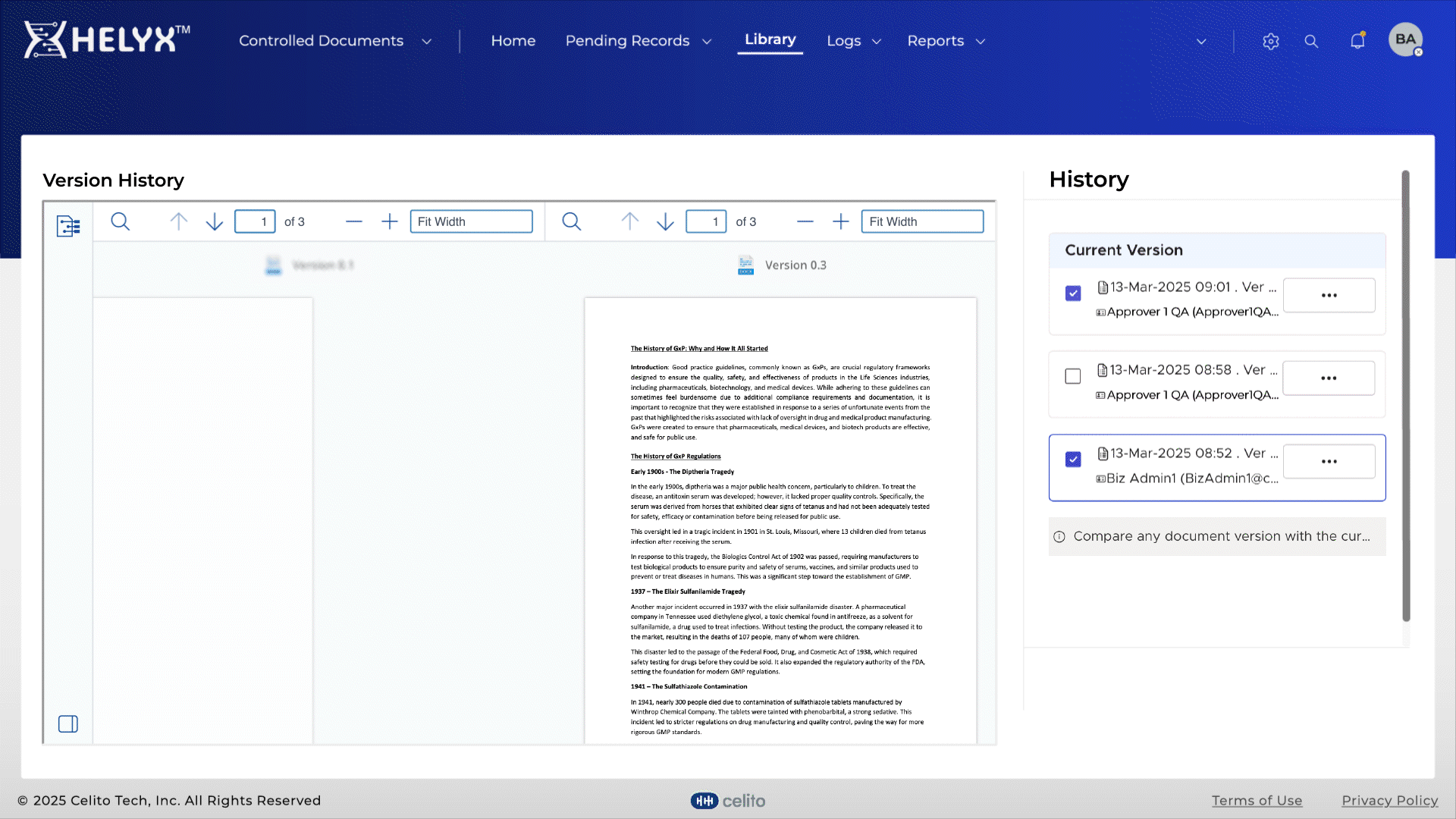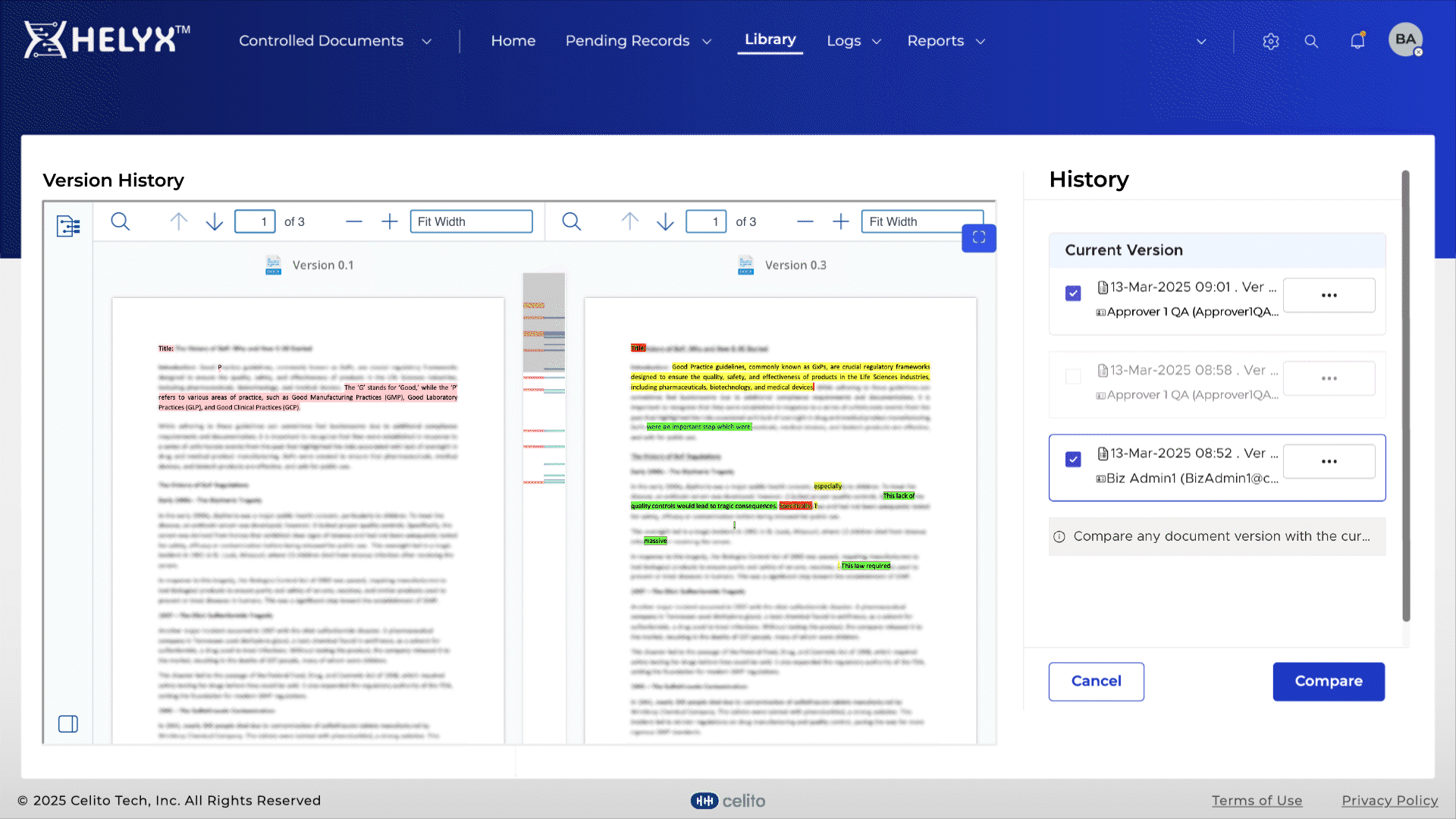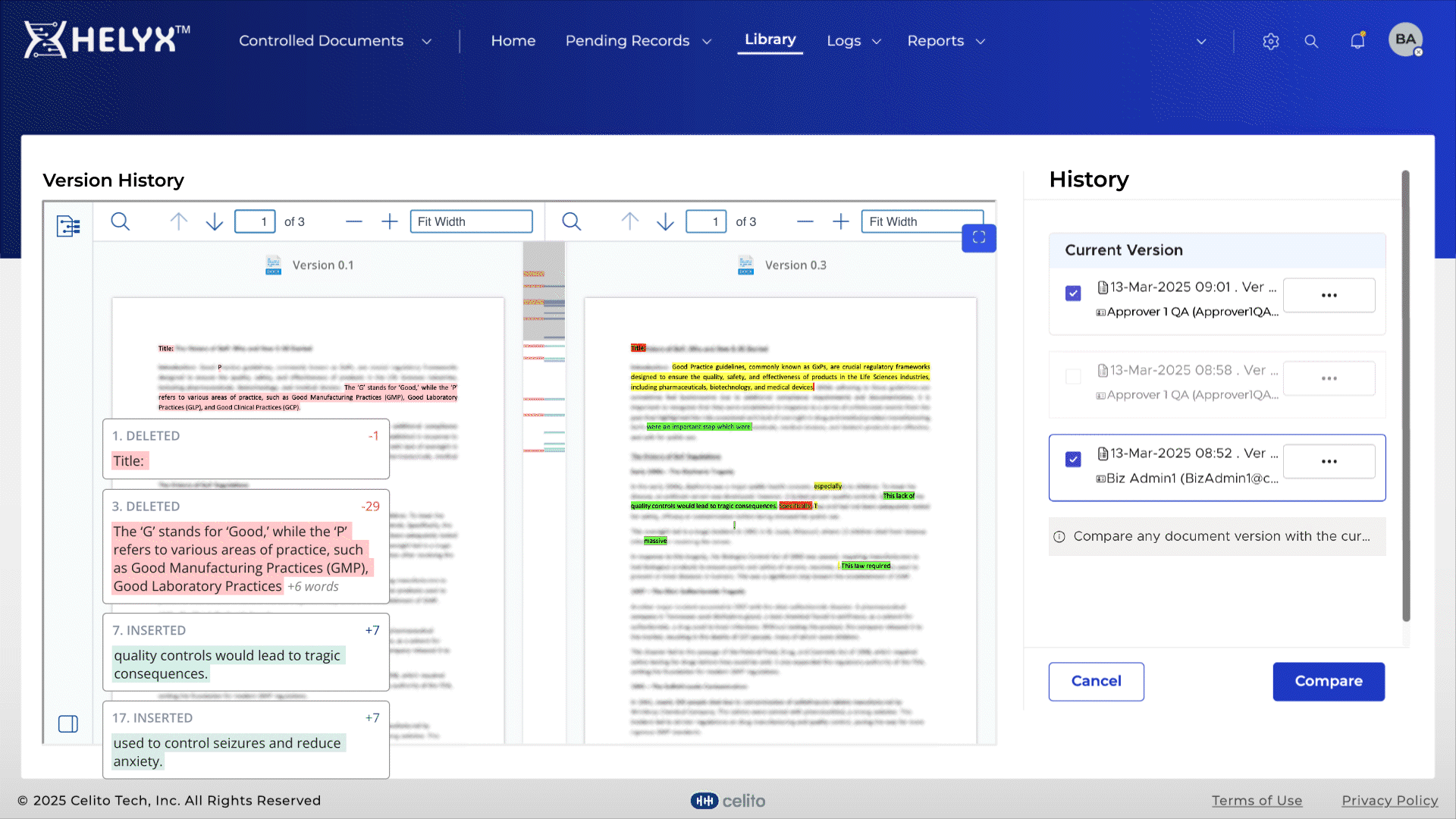
Quickly identify changes between versions to speed up reviews and approvals.
Enable teams to collaborate on documents in real time using Microsoft 365 tools.
Delegate tasks to users for efficient workflow management and timely completion.
Assign role-based permissions so only authorized personnel can access, edit, or approve documents.
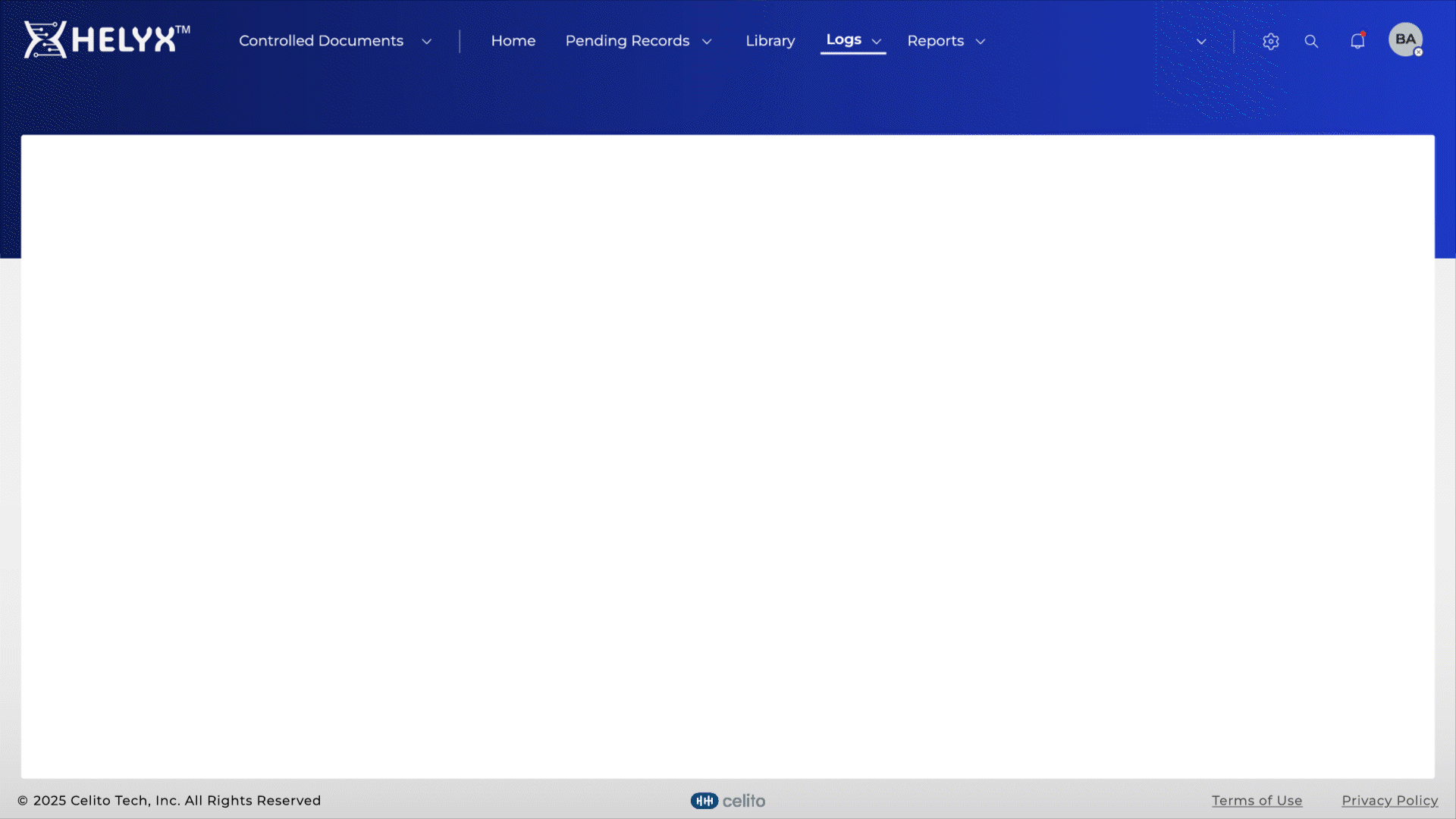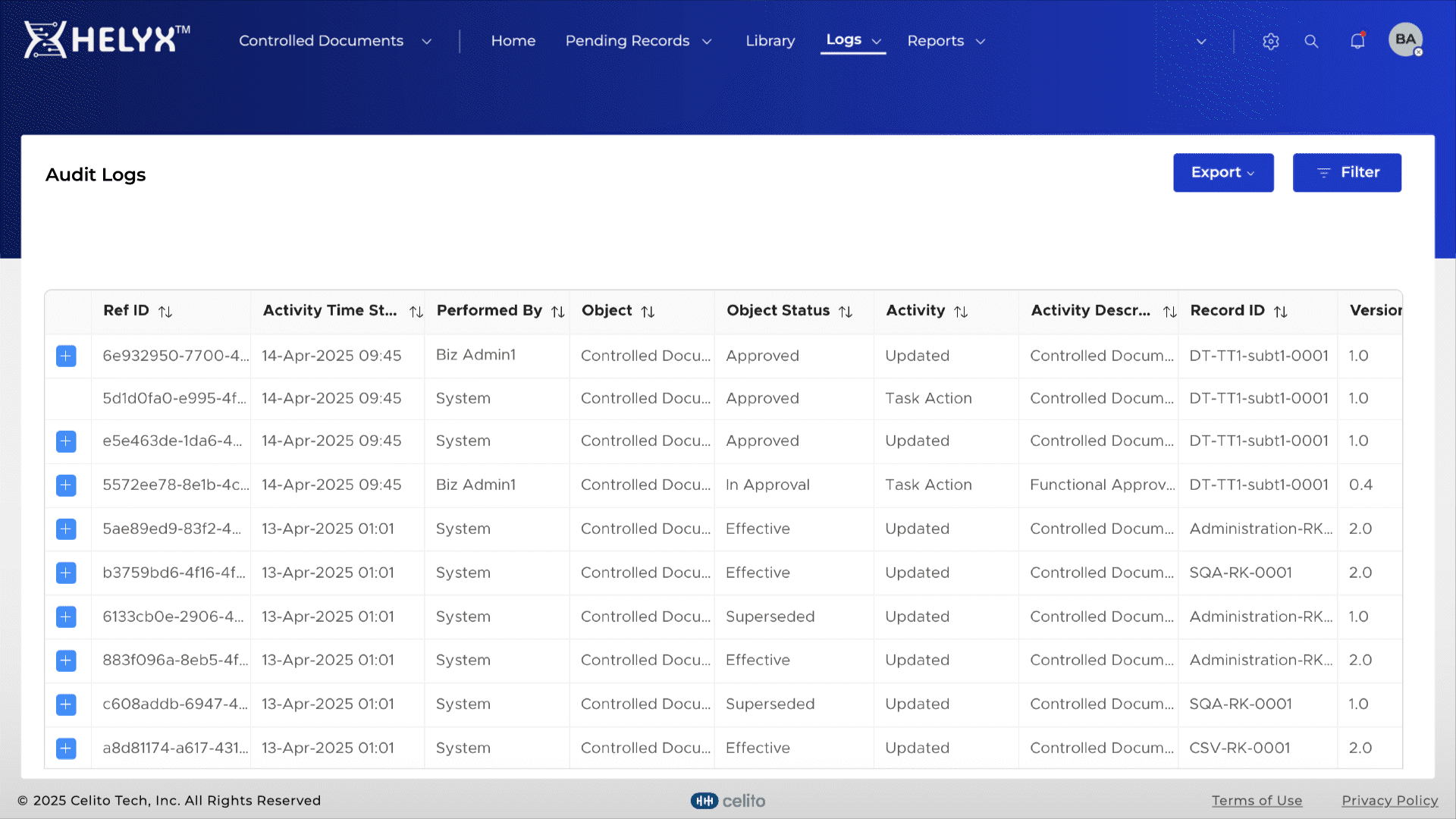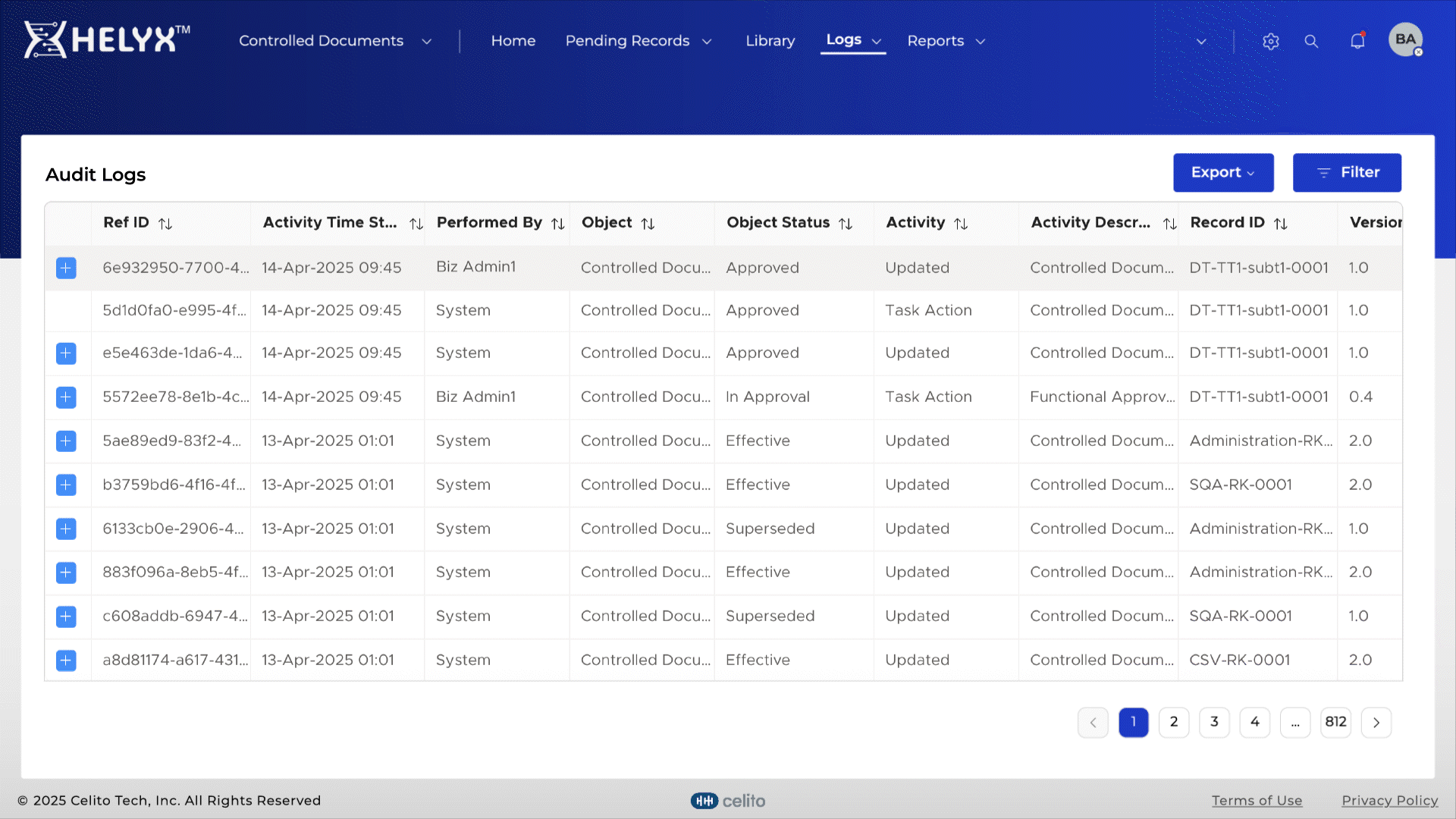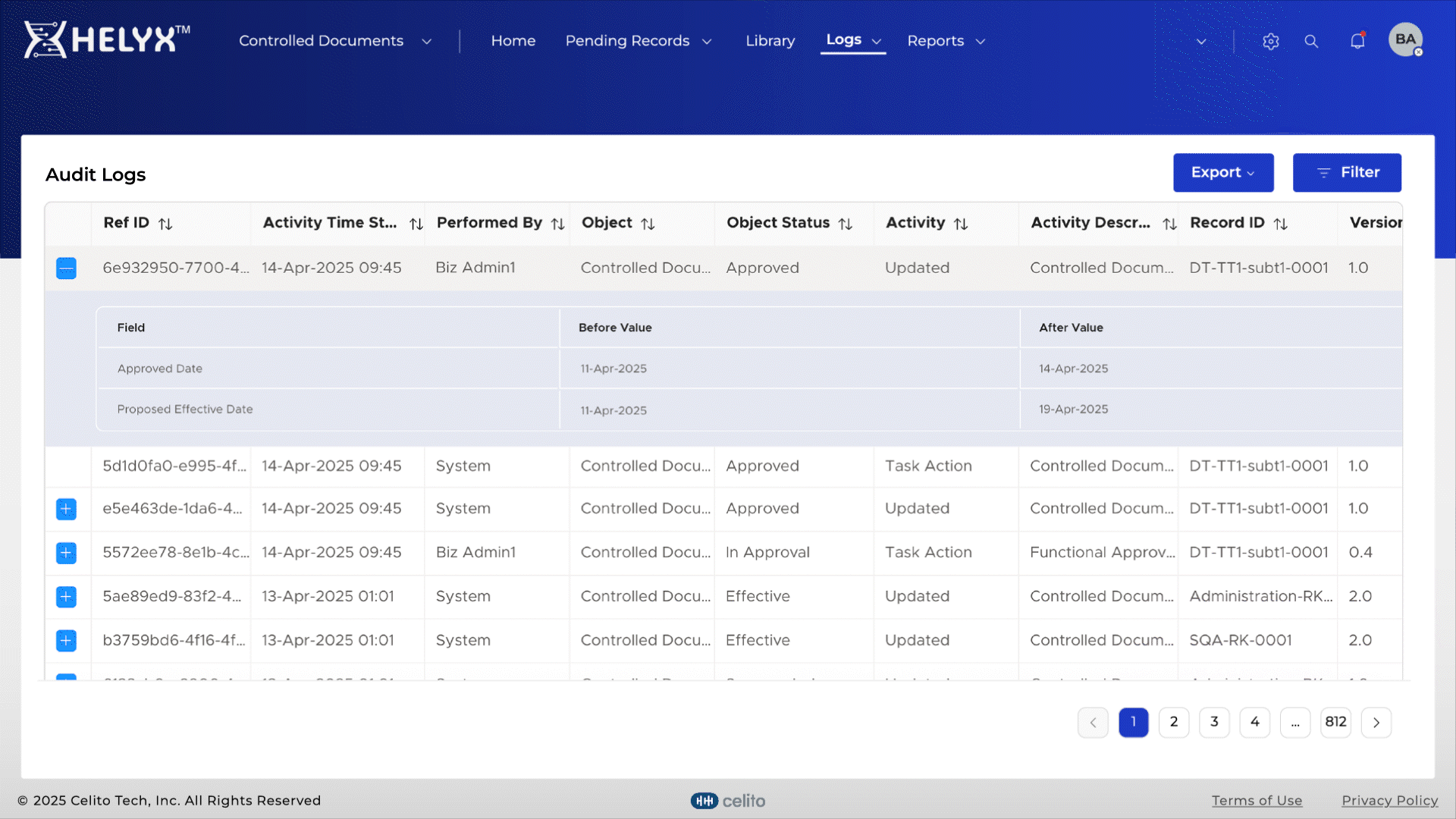
Automatically log actions related to document creation, approval, and distribution.

Built on Microsoft Azure with MFA, encrypted storage, and continuous security monitoring.
Explore Helyx's features, benefits, and how it can drive your success.
Request a Demo